Yves here. Ignacio’s review of some of the key elements of vaccine research, development, and approval will hopefully serve as an important primer for future discussions.
Notice that even this basic discussion demonstrates that a full year after Phase III vaccine trials have started is a bare minimum to reach any conclusions about efficacy….yet the media is braying that we might have an idea on Phase III trials starting now by this October.
By Ignacio Moreno Echanove, an epidemiologist
I regret to say this article is less comprehensive than I would like. For two reasons: time and space. I needed more time to review, read and re-read scientific literature to have a clearer picture. Space: a single post is not enough. I will start with the conclusion: Better understanding of the immunology of Covid-19 and Covid-19 vaccines is central. The more effort is dedicated to this, in both clinical and pre-clinical research, the faster we will get to better treatment and vaccine solutions and less risks will be assumed. Now let’s see what’s going on:
How Good Does a Vaccine Have To Be? When Might We Have One Approved?
First and foremost, before deploying any vaccine on a mass scale, apart of efficacy results, it must have been shown to be safe. In case that there are some safety issues but the protective effect is overwhelmingly beneficial so as to view those risks as a toll we can collectively/individually pay, these should be carefully analysed, described, quantified and communicated to the public before approval.
One of the biggest mistakes would be to approve of a vaccine only to later see more frequent or more severe adverse effects than had been anticipated. Rare adverse events occurring at about 1:20.000 or lower frequency is something that we should to have in mind as a possibility once we have an approved candidate.
The WHO provides general guidelines (2004) for vaccine evaluation/development. It has to be noted that because we are talking here about a novel virus and antigens in all cases, except the BCG candidate, massive trials will be starting with nearly zero previous information even if we can count with experience on SARS1.0 and MERS trials (never scaled up to Phase III) that might give hints but not assurances on vaccine development against Covid-19.
Regarding clinical trials, Phase Iis a safeguard trial done with a few individuals (10-20) to check that the candidate is safe enough for trials with more volunteers. Being in a hurry, some candidates are running directly with so-called Phase I/II trials. So far, such acceleration has not been seen as problematic. So far.
The objective of Phase II (about 100 to few hundreds of subjects) is to characterize the immune response that the vaccine provides and decide if it looks good enough to proceed with Phase III.
Before starting Phase III all considerations about safe manufacturing and scaling up should/must have been settled (I wonder if this was the problem with delays in Moderna vaccine Phase III, but if so, they have just resumed to Phase III).
Phase III (many thousands on subjects, the larger the trial, the shorter the duration, but also depending on the rate of spread when and where the trial starts). Some selection of racial and age cohorts will be necessary given the known information. Phase III is to assess the efficacy of the vaccine, so during Phase III both, placebo and vaccinated subjects, will be naturally challenged in the normal epidemic evolution and tested to see how the vaccine provides immunity/protection against the vaccine. Forced challenging (as in deliberate exposure to Covid-19) has been proposed to accelerate development . As you can imagine, this proposal is the subject of bioethical questions with no easy answer. I think there is a Chinese candidate being tested among the militaries probably with forced challenge.
So far, this is a simplified overview of the clinical trials. Let me introduce some definitions from the WHO document that will help us to understand results better. I will focus only on definitions relevant to the evaluation of the vaccine and set aside those related with the experimental design of the trials.
Adverse reaction: A response to a vaccine that is noxious and unintended and that occurs at doses tested in humans for prophylaxis, or during subsequent clinical use, following licensure. The term adverse reaction is usually reserved for a true causal association with a drug or a vaccine. Tolerability to adverse events has to be defined.
Booster vaccination: Vaccination given at a certain time interval (at least 6 months) after primary vaccination in order to induce long-term protection. [Note that primary vaccination can consist in one or more doses repeats in a short span]
Geometric mean titre: Calculation of the average titre (of antibodies reacting with the antigen or a relevant part of it) for a group of subjects by multiplying all values and taking the nthroot of this number, where n is the number of subjects.
Immunogenicity: The capacity of a vaccine to induce antibody-mediated and/or cell-mediated immunity and/or immunological memory. [This is where I am skipping over a more comprehensive discussion]
Potency: The quantitative measure of the specific ability or capacity of the product to achieve a defined biological effect. For instance, quantitative virus neutralization assays. [This is of course all important for those vaccines based on the SARS CoV 2 Spike protein or epitopes therein. Neutralization titres (VNTs) usually correlate very well with immunity and vaccine efficacy]
Reactogenicity: Reactions, either local or systemic, that are considered to have a causal relationship to the vaccination. [As we will see, vaccine candidates using SARS CoV 2 Spike protein result in relatively high reactogenicity. Not severe but yet an issue and if a vaccine candidate based on the Spike protein is finally approved, we will have to accept some nasty symptoms shortly after vaccination. This will have to be carefully studied in all age cohorts. It has some potential to be problematic.]
Serious adverse event: An event occurring in connection with the clinical trial that results in death, admission to hospital, prolongation of a hospital stay, persistent disability or incapacity, or is otherwise life-threatening. [When a serious event is identified it will force prospective monitoring in the subjects of the trial. Uncommon serious adverse events might not be detected during the trial so it is advisable a follow-up for these if the candidate is approved.
Rare events: T hese are usually detected retrospectively. ‘Common’ adverse effects are defined as those occurring at ratios between 1:100 and 1:1000 while less common but not necessarily rare would go to about 1:10.000. Below this frequency we could deem as more or less rare events. Detecting and monitoring adverse effects is one of the most difficult challenges during Phase III trials and that is why risk assessment in preclinical trials is important.]
Seroconversion: Predefined increase in antibody concentration, considered to correlate with the transition from seronegative to seropositive, providing information on the immunogenicity of a vaccine. If there are pre-existing antibodies, seroconversion is defined by a transition from a predefined low level to a significantly higher defined level such as a four-fold increase in geometric mean antibody concentration.
Vaccine (protective) efficacy: The reduction in the chance or odds of developing clinical disease after vaccination relative to the chance or odds when unvaccinated. Vaccine efficacy measures direct protection (i.e. protection induced by vaccination in the vaccinated population sample). Vaccine efficacy is calculated according to the following formula:
Where Iu = incidence in unvaccinated population; Iv = incidence in vaccinated population. [Importantly the end-point is defined as ‘clinical efficacy’ but the endpoint definition(s) of efficacy will be made according to outcomes: absence of infection or immunity, reduction of virus shedding, mild disease, preventing development of lung lesions, hospitalization required… Immunity would be the most stringent definition. When the trials include placebo treatment (an inactivated vaccine or an alternative vaccine) and double blinding these are called superiority trials and will demonstrate if the vaccine gives superior results to the placebo treatment. The estimate of efficacy will be defined by statistical estimates with, for instance, quantitative RT-PCR diagnostics, virus shedding analysis, visualization of lung lesions by CT scanning etc. Being this a new disease, we cannot yet resort to ‘surrogate’ indicators such as plasma VNPs.]
Vaccine failure: The onset of infection or disease, biologically confirmed, in a subject who is supposed to be protected, following completion of age-appropriate immunization as recommended by the manufacturer.
The WHO guidelines state that pre-clinical and laboratory evaluation are pre-requisites for clinical evaluation of vaccine candidates but this step has been skipped in some cases. The rationale for this is that, apart from the rush we are in, the coronavirus itself was not used to make the vaccine. If a vaccine candidate consists on inactivated SARS CoV 2, pre-clinical stage cannot be skipped. The primary objective of the pre-clinical evaluation is to demonstrate that the candidate is suitable for testing in humans. This should include indicators of safety in both in vitro assays and in animal models. Yet, pre-clinical studies provide more than that and are very useful to study risk of known adverse effects.
For Covid-19, to my knowledge, there are three different animal models available: murine (mice, rats, hamsters), ferrets, and a few non-human simian species being the latter the models that more closely resemble human Covid-19 infection. Toxicity of the vaccine has to be evaluated in these models and this includes determination of safe doses, need for repeated doses, tolerability, potential to induce antibodies that cross-react with human tissues…
The WHO document doesn’t explicitly mention the possibility of ADE (Antibody Dependent Enhancement) and/or VERD (Vaccine-associated Enhanced Respiratory Disease), and this should be considered a must in preclinical phase. The FDA has released guidelines for Covid-19 vaccine developers, with non-binding recommendations. One of the pre-clinical key factors listed that should be addressed is VERD. ADE is not mentioned though it can be the mechanism behind some cases of VERD. An excellent discussion on how to address this problem can be found in this paper, not good reading for the immunology-naïve, but for us, the common people, it might be enough to highlight the main conclusion:
Although the development of vaccines and therapeutics for SARS-CoV-2 remains urgent, we must proceed with caution, using the full armoury of vaccine and protein design tools at our disposal to rationally minimize the risk of ADE.
Remember these phrases ‘proceed with caution’, ‘use advanced tools’. Looks like we are not doing enough of this.
Preclinical assays will also inform on vaccine potency, immunogenicity and efficacy in such animal models. Preclinical trials also help with the selection of adjuvants, additives, formulations and other vaccine-specific information. Since the pre-clinical pre-requisite has been removed in several vaccine candidates several scientists have complained this might be a mistake. It could indeed be a mistake.
Given that this is a new and rapidly spreading disease, efficacy testing Phase III trials should be large. Somewhere between 10.000-50.000 individuals and the follow-up would take 1-2 years though some conclusions on efficacy might be obtained in about a year. If some kind of protection is seen it is crucial to check the duration. So, if at least a 6-month duration of protection –before and in case a booster vaccination is seen as necessary after 6 months or later– is a pre-requisite for approval this means that first results won’t be seen until about one year after the start of Phase III trials.
Recruiting volunteers, vaccinating them, time to full development of immune response, challenging of subjects through the natural course of the epidemic, more than 6 months monitoring after immune response, and all the work associated mean that 1 year is a bare minimum for results. Everybody is trying to be optimistic with the timelines but it will be challenging to have something approved before the end of 2021. This could be accelerated with forced challenging, but results would be obtained with age cohorts that are not representative of the most susceptible part of the population. Given the current state of the epidemic, the US it is now one of the best places in the world to start Phase III trials.
Another way to accelerate this would be relying on wishful thinking after some months of promising results.
Phase IV studies (after approval) are basically safety evaluation studies and should be mandatory for Covid-19 vaccines given the uncertainties mentioned. Pre-exposure cohort studies or secondary attack-rate studies will also be needed given the high attack rate of Covid-19.
Evaluation of vaccine efficacy. Given the epidemiological characteristics of Covid-19 (high attack rate in vaccine-speak), depending on the measured efficacy, the observed seroprevalence at the start of the vaccination program, and the results of pre-exposure cohort studies it will be decided what is the coverage needed in the population. The higher the efficacy, the lower the coverage. According to a study based on computational models (Graduate School of Public Health and Health Policy, New York City, NY, USA) applied to Covid-19 epidemics…
…‘to either prevent or largely extinguish an epidemic without any other measures (e.g., social distancing), the vaccine has to have an efficacy of at least 70%.’
Let’s keep in mind this 70% efficacy as a reference for the future.
I will end this section underlining a fact that can be important about fast-track candidates. These candidates were designed very early in the pandemic, as soon as the SARS CoV 2 genome sequence was published. so their respective designs didn’t have in consideration the knowledge on the disease that has been accumulating ever since. Speed, in this sense could be an advantage but could well also result in high failure rates. Late candidates might benefit from better knowledge on the immunological and pathological aspects of the disease, as well as mistakes of the speediest. Just let’s hope these mistakes are not too dramatic.
Current Timelines
Now let’s take a look at the vaccine race as preliminary results are being announced or published by some candidates. Next figure from the GAVI site gives a geographical snapshot on the current state of the race with candidates already in clinical trials.
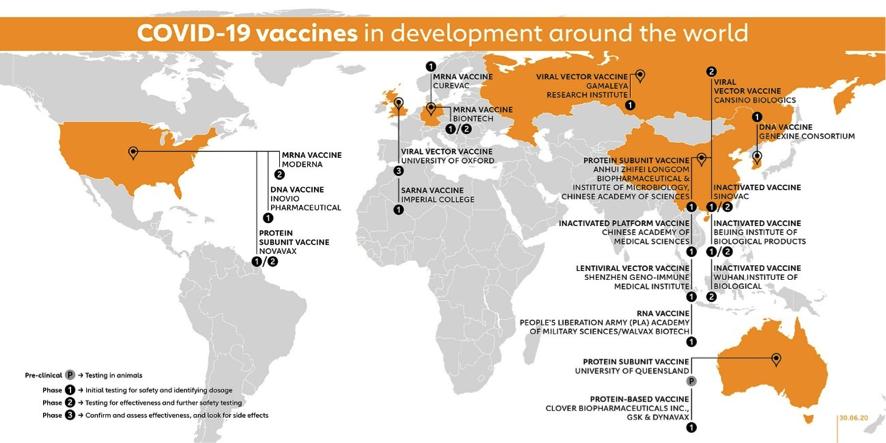
The figure above includes 10 vaccine candidates already in Phase I, 4 candidates in combined Phase I/II trials, 2 candidates in Phase II and 3 Candidates in Phase II/III (Moderna is already for a total of 18 candidates in clinical phases. Impressive!
The University of Oxford-AstraZeneca candidate (AXD 1222, UK, Adenovirus vector) was first to announce a Phase II/III trial in May 22ndwith about 10.000 volunteers in the UK, and including small children/elder cohorts. It is said to be expanded with trials in Brazil, South Africa and the US with now more raging epidemics. It had already showed preclinical results Rhesus macaques and additional preclinical trials are ongoing.
This project also aims to soon start controversial challenge trials in which healthy participants, vaccinated or not, will be ‘artificially’ challenged with SARS CoV 2. This might accelerate efficiency results but also rises serious concerns about safety issues that could backfire later. Australian commenter Hilda Bastian highlights that this also increases what she calls the “activism risk factor” or vulnerability to deliberate doubt- sowing on vaccines. I strongly recommend reading her posts in full.
Moreover, given that 3 out of 6 macaques vaccinated with this candidate and then challenged with SARS CoV 2 showed symptoms of respiratory distress one wonders if this could be the best candidate to try forced challenging with human subjects.
The Wuhan IBP-Sinopharm CNBG is a 100% public project (China, Inactivated virus) that in June 23rd announced start of Phase III in the UAE, and has undergone mandatory preclinical studies given it contains virus.
Similarly, SinoVac Life Sciences (China, inactivated vaccine) has published preclinical results, has ongoing Phase I/II trials and planned to start Phase III in Brazil in July with nearly 9,000 participants according to NIH site for this trial with results expected in October 2021.
Moderna candidate (mRNA-1273, US, mRNA vaccine) is set to start Phase III trials on July 27thwith 30.000 participants. No preclinical studies done or planned.
CasSinoBIO (Ad5-nCoV, China, adenovirus vector) has undergone Phase I/II trials and in 29thJune was announced it had received “military specially-needed approval” and this means approval limited to military use in China for at least one year. They announced on July 11th talks for Phase III trials with Brazil, Russia, Chile and KSA and expect to enrol about 40.000 subjects.
The Pfizer-BioNtech (BNT 162, Germany, mRNA) candidate has just published Phase I/II results with one of their variant candidates (1b) and has also showed preclinical results. The developers plan to start Phase III later in the summer enrolling about 30.000 subjects in the US. BioNTech CEO believes hat BNT162 could be ready for approval by the end of the year. As I see this, the 6-month protective duration prerequisite could only be fulfilled if Phase I/II subjects are ‘artificially’ challenged later in the year.
So, there are 6 candidates already in or about to enter Phase III trials. One wonders if this is the result of rational thinking or if we are running all candidates into Phase III trials like a run of beheaded chickens. Time will have a say on this.
Other players that might go in relative short times to Phase III include Inovio (INO-4800, US, DNA vaccine) that has announced “positive interim Phase I data”, and Novavax (US, protein subunit vaccine) whose stocks had gone through a 3500% rally between May and July and is expected to publish results of Phase I clinical trials anytime soon. Novavax looks to be now on dive-watch in case the results fall short on expectations. Brokers are learning what VNTs mean but I am not quite sure if they can interpret the results correctly particularly if already published numbers can be readily compared.
We don’t know how this will end but billionaires, as well as the corresponding losers, are made along the way no matter the final results. Among other mRNA candidates, CureVac (Germany, mRNA), is expected to show first clinical results by September, has announced promising pre-clinical results.
Now I think I have touched a theme that is central to Naked Capitalism which is about the discussions on how markets operate and if we should believe on those neoclassical pontifications on ‘rational market expectations’ versus market failures related with not so rational expectations. It looks like, and we are seeing now this ‘in vivo’, these so called ‘rational expectations’ frequently work with much less than perfect information, for instance VNTs estimated in a Phase I study. There are risks associated with vaccine development, that are the result of truly rational thinking, which have not yet been resolved and aren’t considered in such Phase I/II results. In the meantime, markets might show wild gyrations on the basis of data that are or should be known to be inconclusive/insufficient. Imagine that some vaccine candidates fail to be marketed, –most will do so–, but on the way, billionaires are created to the expense of many others. Is this a rational market or something that better resembles lottery winners? I leave this as food for thinking.
The newsrooms are all alert on the preliminary results and announcements being issued this summer on clinical trials. It is tempting to do a comparative analysis trying to identify who among the front runners is showing better results but because these are all preliminary, I think this would result highlighting the selling points that each candidate wants to make depending on their particular results. Also, because some candidates have skipped preclinical studies and because ‘surrogate’ indicators using equal materials and methods (the best would be a single or a few coordinated labs doing this with standardized protocols) do not really exist, conclusions could be misleading.
I will leave for a posible future post a discussion of the immunological aspects of the vaccine and the reported results if I manage to become confident in what I write. So far, I have seen information on preclinical studies which is scant, involving very few animal subjects (particularly non-human simians), and in some cases with insufficient immunological analyses. The same caveat about immunological studies can be extended to published Phase I/II results. There might be better studies that haven’t been made available to the public or ongoing efforts to complement those. I don’t know, but not knowing I stick to the precautionary principle.


“The University of Oxford-AztraZeneca candidate….”
I believe the spelling should be “AstraZeneca”. If so, that corporation has engaged in criminal activities: “In 2003 federal officials announced that AstraZeneca had pleaded guilty to criminal and civil charges relating to the illegal marketing of the prostate cancer drug Zoladex. The company agreed to pay $355 million, consisting of $64 million in criminal fines, a $266 million settlement of civil False Claims Act charges, and a $25 million settlement of fraud charges relating to state Medicaid programs. AstraZeneca, which agreed to enter into a corporate integrity agreement with the Inspector General of the U.S. Department of Health and Human Services, had been accused of giving illegal financial inducements such as grants and honoraria to physicians.”
https://www.corp-research.org/astrazeneca
This then raises the question of how many corporations that have paid criminal and/or civil penalties are involved in vaccine studies.
In the AstraZeneca case, should a criminal organization be given any public money to do research? Should it be involved in any of these critical studies at all? It certainly raises a lot of questions about the institution of Medicine and the University of Oxford when they do business with criminal organizations.
Concerning medical research in general and the problem raised above is this item dated July 28, 2020:
“BOSTON – The former Chair of Harvard University’s Chemistry and Chemical Biology Department was charged today in a superseding indictment with tax offenses for failing to report income he received from Wuhan University of Technology (WUT) in Wuhan, China.”
https://www.justice.gov/usao-ma/pr/harvard-university-professor-charged-tax-offenses
Therefore, it would seem that any discussion or analysis of vaccines should address the criminogenic environment in medical research.
Will correct, I did a light edit but missed that. And more important, thanks for the background.
Sorry for the typo. Regarding the charges with tax offenses it was weird to find that position as ‘strategic scientist’ for the U. of Wuhan which I guess has not so much to do with real science but lobbying and network building.
Marcia Angell has covered the crookedness of big Pharma for years. See NY Review of Books…for her many publications
This is undoubtedly a factor in the public distrust of the vaccination effort. For that reason alone, not to mention the morality, all bio companies should adhere to strict standards and verification. I’ll believe it when I see it.
Thanks so much for this, an excellent and informative overview. As so often, NC kicks ass over the media when it comes to technically sound analysis.
The more I read about this topic though, the more I fear that the goldrush for whoever gets their vaccine accepted first could result in a cure worse than the disease. Good science will always be a casualty when so much money is at stake.
Or it might be somehow better than the disease but yet suboptimal. For instance it could be protective but fail to avoid disease spread.
As cancer specialists often point out, its far more profitably to have drug treatments that prolong lifespan for several years than it is to have a drug that rapidly kills the tumours.
Disease spread is not as much an issue if it effectively neuters it to a less deadly form. Like the flu vaccine, the goal isn’t to eradicate it as much as it is to allow it to not disable or kill you if you catch it.
You are counting that through vaccination you are protecting everybody. Vaccinating 60-70% would still leave many vulnerable.
Thanks Ignacio. Great info once again. I notice that China has chosen inactivated virus vaccination production whereas the US, Germany, and the UK seem to like mRNA vaccine technology. I’ll be looking for your next post on Immunogenicity because I’m in a tizzy that immunogenicity will be entangled with the risk of possible very long term negative effects if mRNA vaccines cause some little cellular glitch and go occult only to emerge 2 generations hence. And blablablah. Those things would be virtually undetectable, if they existed at all, I’d assume. And your tidbit of the risk of ADE/VERD adverse situations of actually enhanced disease sounds like a nightmare. I just wish we would put vaccines on the slow track and put medicines to treat Covid on fast tract. Medicines with antidotes themselves.
Thank you for the good work!
My comment about Ignacio’s comment about a typo not making it into the comments to his article ended up in the wrong place. mea culpa. Sorry. The link takes you to his article but not his comment.
This is really excellent background, Ignacio. Thank you. I would like to ask if you think it is possible that Covid 19 is not a candidate for a vaccine. Is it possible that a cocktail of medications (more expensive, for sure) would be a better approach? Also, do you know if any efforts were made to develop a vaccine for SARS or MERS?
Efforts for SARS & MERS vaccines have been done but because these diseases got controlled none of the candidates reached expensive Phase III trials and none was approved. ADE was seen in a few instances. So…
Regarding the disease as a ‘vaccine’ aka herd immunity it is known seen as highly problematic because disease- associated outcomes (most well reported here) of course and because immunity even for as short as 6 months is far from guaranteed.
Yes! Thanks! Great summary.
So bottom line, we shouldn’t expect anything serious in terms of safe and effective until at least summer 2021?
And durable for at least 6 months.
Ignacio- Thanks for this important primer. Do you have any information about the Russian vaccine project supposedly about to be approved for use?
Let’s pretend we’re in Vegas. I’m betting the house there will be no effective vaccine ever for this novel virus. Effective is the operative word here. There will be vaccines developed by huckster pharmaceutical companies and they will range from outright ineffective to downright deadly and keep in mind they are now legally immune from liability thanks to legislation that will be passed into law shortly.
I also believe that we have entered another era of pandemics meaning in the next several decades we will see one pandemic after another, each prompting vaccine efforts. There is no capacity for all of that research and never will be. Couple the emerging era of pandemics with all the other shit that’s hitting the fan, and we can quickly conclude that even the most clever disaster capitalists cannot keep up with the flourishing opportunities to exploit tragedy.
As an investment advisor, I suggest you go long on euthanasia drugs. Considering the incredible suffering to come, the humane thing to do is to allow the unwashed, and washed too, to exit this world in dignity on their own terms. There is no good reason for billions to needlessly suffer when the fate of humans, let alone civilization, is pretty much written.
The Suicide Tourist
I’d love to have a stockpile on hand…for the right moment. It would give me a great deal of peace of mind.
Yes it looks that way Plague. On the other hand, we could monitor them properly for once and usher in the long anticipated golden age of capitalism. Monitor being the key word for the future.
Oh yes, because ‘downright deadly’ vaccines are really oh so common (I’m aware of precisely *one*, a dengue vaccine that triggered ADE, and it was withdrawn in a panic as soon as this became evident: dengue is known to be an unusual disease in which subsequent infections are worse than the first in any case: there is no evidence whatsoever that this is true of any coronavirus whatsoever: this is rare enough that tens of thousands of pages have been written on this one event).
Enough of this lunatic conspiracy theorizing. If you must insist that everyone is driven purely by naked self-interest, note that the people working on these vaccines and the CEOs of their companies are seriously affected by lockdowns and other counter-covid measures just as we are: they have every selfish inclination to make a vaccine that works, and works well. (Because *of course* scientists go into vaccine research to make money! They’re rolling in it, it’s far more profitable than, say, deciding to become a quant).
Having been one of those virtue-minded pharma scientists, you have to remember we were not in charge. We didn’t make the ultimate decisions. That fell to our management ‘betters’.
I believe this is pertinent and relevant. In America at least, with the election of Trump to the Oval Office, we have entered an era, yet again, of Caveat Emptor (let the buyer beware). Applying that to a vaccine spells disaster and if the vaccine is mandatory, the humanitarian disaster could be greater than what the disaster the virus itself can produce.
Don’t get me wrong, I hope upon hope a successful effective affordable (I would argue for it to be successful and effective it must also be affordable) vaccine will be developed, but knowing human nature and human organizations and extrapolating the corruption trend curve, it doesn’t look promising despite the propagandistic hype.
Also, the Dengue Fever vaccine was not the only deadly vaccine on record. There was also this and the historical parallels are significant. It’s highly instructive, to say the least. The perfect storm is brewing. All the ingredients are in place and Donald Trump us just the man (if he can be called a man) to bring them together because, afterall, it does appear to be his goal to maximize the number of deaths from this virus and a bad vaccine would do the trick.
https://www.washingtonpost.com/history/2020/04/14/cutter-polio-vaccine-paralyzed-children-coronavirus/
Go long on handbells, too. We’ll soon be back to medieval scenes of the plague-infected walking the streets and ringing them while shouting ‘unclean’ to enforce social distancing.
One thing I wondered about which has been mentioned on NC in passing, is so-called ‘first mover advantage’ for vaccine development.
This would be the idea that a lot of money is to be made for a company or group of scientists who push out the fist ‘more or less OK’ vaccine and convince enough people that it works well enough to warrant maximal deployment.
While this might be the case if the vaccine indeed works, if it doesn’t this would surely backfire on the company involved, no? What would be the gain for pushing out a not so effective vaccine but beating the other vaccine developers? Or is that a longer-term problem for whoever is left at that company to clean up the mess?
Related to this, many countries including most EU countries and China have signed up to a ‘vaccine pool’, which I believe is some sort of pledge to not patent their vaccines. Notably, the US, UK and I believe Switzerland have not. So if the first ‘good enough’ vaccine is from the UK (e.g. Oxford), they can charge whatever they want for X years, while if one of the Chinese ones is first (e.g. SinoVac), other countries would presumably be free to make their own generic version of it. Is this correct, or have I misunderstood?
Since we cannot predict how these will work and if there are more or less mild or severe effects and how frequent these could be and if these will be detected during Phase III it is still soon to answer your question.
I hope that anything serious is readily detected during the Phase III studies and I hope it isn’t too serious and too frequent but some actors are going as if these possibilities do not exist and play with too premature approval dates.
I hope so too. I think there can be a lot of damage done by a bad vaccine released to soon–health damage obviously, but also damage to credibility of future vaccines.
By the way Ignacio, many thanks for your tireless coverage & explaining these complex matters–and to NC for giving him the platform. Truly the best place online to stay ahead of the curve.
Please add my thanks to the pile!
If i may ask, are there noteworthy differences among the types of vaccines being tested that are significant in terms of how their testing should go? (How long to test, tougher criteria for success in any areas, more care to guard against serious adverse effects, and so on?)
There are differences. in compositions, and both quantitative and qualitative differences in immunogenicity. Vaccines that consist on inactivated virus must ensure full inactivation in all batches. There are also differences in the dosages needed to show immunogenicity. If I can I will try to summarize available information including other candidates that are behind the front runners.
Thank you (in advance) again!
I don’t have much money, but I donate monthly to this blog and it is money very well spent. I urge others to do the same. You won’t get this level of reporting from other sources.
‘the vaccine has to have an efficacy of at least 70%.’
Isn’t this about the same percentage for so called herd immunity? I have read figures for this from 60% through to 85%. So it seems that a vaccine as planned here appears to replicate a herd immunity approach but I am betting that the pharma companies are hoping for seasonal shots for any vaccine so that they can dump doing any research on an actual cure.
Many thanks to Ignacio for all his hard work here. It is a much more complicated situation with vaccines than I thought and this article highlights the difficulties to be overcome. Muchas gracias.
I am not sure if that efficacy is measured in terms of absolute immunity. So far, preclinical trials suggest that immunity is a tall order for Covid-19. Is macaques the vaccine candidates that have been tried do not provide immunity. Challenged macaques are readily infected but show better outcomes particularly in their lungs.
Depends on what you think the R0 with no protection is. Some later research suggested over 5. That means you’d need immunity in over 85% of the population.
https://wwwnc.cdc.gov/eid/article/26/7/20-0282_article
Would it be accurate to say that those most at risk of Covid-19 could not take any possible vaccine as the “pre-existing condition” would leave them potentially compromised by the vaccine?
Also, perhaps this is beyond the scope of this most informative article: are the dangers compounded by rushing a vaccine, by the fact that there is so much skepticism in America that say, even if it proves safe (if not necessarily effective), if only 40% of the population takes it, is it conceivable or even likely that that could be even more deleterious and destructive, spreading the disease even more, rather than merely approaching it with the current distancing and mask directives?
I get the sense maybe that the emphasis on “speed” is not about saving lives but saving the economy (equities primarily), which if that is indeed the true but denied premise then the odds are whatever vaccines are rushed to market are more harm than good?
Vaccines are becoming a deus ex machina. There is nobody who wants a safe and efficacious vaccine more than me – getting covid is a death sentence for me (and millions of others).
But no matter how much I want it, we won’t get it as soon as we want it, and it is possible, that we will never get one. (or we will get one of dubious efficacy – man, what a clusterf*ck that will be)
I don’t know how many times when I was at FDA there were articles about progress toward a HIV vaccine, or progress toward cancer cures. Those cures are about as common as those flying cars that were going to happen…
The era of infectious diseases being tamed is a second out of the week of human existence. We TAKE IT FOR GRANTED – it is not inevitable.
Every day, hundreds of trillions of microbes are evolving with one goal – to make you their lunch. I only wish we would stop slathering ourselves in gravy and get off the dinner plate…
I think people are leaving out 2 very important items. First, vaccines are being developed all over the world. And some are bound to be used before the USA. China may be doing so already. And second, there are huge populations at such high risk, that many will take that first chance. e.g. HC workers of all sorts. Nursing home and chronic care facility patients.
Then the 3rd which I almost forgot, and favorably muddies these waters. That being monoclonal AB treatments. These will be out sooner, and may well convert the bulk of Covid 19 to non-lethal cases.
https://www.usnews.com/news/health-news/articles/2020-02-03/hiv-drug-costs-soaring-jeopardizing-effort-to-end-epidemic
You miss that the longest immunity to any major coronavirus is only 34 months, for MERS, which also to make the #s easy, also has a 34% case fatality rate. It’s only 6 months for the common cold. A vaccine may not confer much immunity.
As an epidemiologist, what are your thoughts on Dr Harvey Risch’s recent commentaries on HCQ in COVID-19?
You have to talk about the craziness that is Moderna.
Operation Warp Speed is the US vaccine program, and we seem to be most of our ‘chips’ on the Moderna candidate, and it seems… well… read this first: https://en.wikipedia.org/wiki/Moderna
a) The Moderna candidate is a mRNA based vaccine. There are no existing, approved mRNA based vaccines. There have been many attempts, but all of them have had major problems. Also, by design all of them will produce antibodies, so presence of antibodies isn’t an indication of success.
b) Moderna has not been peer reviewing or releasing their full results. They have been cherry picking cases and releasing data on them.
c) Moderna is run by a bunch of hedge fund types. Normally… not necessarily a problem, but these guys are making a lot of $$$ by releasing partial results then selling stock in the market.
d) One of the board members for Moderna quit… to become chief scientist for Operation Warp Speed. (Like I said, they got most of the ‘chips’.).
e) The phase III trials of the Moderna vaccine is due to be complete Oct 27th: https://clinicaltrials.gov/ct2/show/NCT04470427?term=vaccine&cond=covid-19&draw=5…. That’s right… their results come out exactly one week before the US elections.
f) All of the other Phase III trials are taking 9-12 months…. or longer. Moderna is only taking 3!!!! Plus their primary and final results are on the same date!
https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines?fbclid=IwAR3AkFuzFh_7fybRUKFxqi2SFJtLrDmpIQeuLY-dLz3CkeL-dMqTF909k64
Either they are truly black swans… or they are getting rich setting up an October surprise.
Excellent! A truly great review of vaccine development and the current status of Covid 19 vaccines in development.
Thank you Ignacio and thank you Yves for providing this to NC readers.
Moderna has performed pre-clinical studies. Their Macaque data was published July 7th:
https://www.nejm.org/doi/full/10.1056/NEJMoa2024671?query=RP
After 2 immunizations (week 0 and 4) ID50 geometric mean titers of 501 in the 10-μg dose group and 3481 in the 100-μg dose group. From the abstract: “Viral replication was not detectable in BAL fluid by day 2 after challenge in seven of eight animals in both vaccinated groups. No viral replication was detectable in the nose of any of the eight animals in the 100-μg dose group by day 2 after challenge, and limited inflammation or detectable viral genome or antigen was noted in lungs of animals in either vaccine group.”
They also measured CD4 and CD8 responses. They measured spike protein specific Thelper classes by intracellular cytokine staining after incubation with peripheral mononuclear cells pulsed spike protien peptides. They found significant TH1 but no TH2. I am a bit confused by this as they got strong antibody responses yet not TH2 which normally drive antibody responses. Is it possible that antibody responses are only driven by tfh cells that are sequestered in secondary lynphoid organs surrounding germinal centers? To me another confusing result is the TH1 response. Specifically a TH1 response in the absence of any CD8 response. They state that no spike-specific CD8 cells were detected but I could find no method used for the assay. If high TH1 responses are present why is there no CD8 response?
So I have much to do to try to figure out these data.
Once again thank you for this post
Ignacio, many thanks for this most informative and revealing post. The linked article discussing mechanisms ADE and VERD was truly frightening — particularly the association of these complications with waning antibody levels, as we have seen in other coronavirus diseases. The saga of the dengue vaccine is illustrative of potential problems. I am looking forward with trepidation to reports of Covid-19 reinfection and its consequences.
This post was a great round-up, Ignacio! Thank you very much! I look forward to your future post, if any, concerning the “immunological aspects.” (And I always appreciate your comments, generally.)
I have seen reports in the English-language Russian press of a vaccine those people are developing but it does not appear to make it to this article.
Because there is zero technical detail.
Many thanks, Ignacio for this information. I always look forward to reading your posts and take away much information which isn’t found anywhere else. You’ve confirmed my suspicions that a vaccine for this virus won’t be available this year. It’s amazing to look back when the Salk vaccine came out – I believe the live virus was used and when the Sabin oral vaccine came out it was a “dead” virus. People were so desperate to take the live virus because polio was such a scourge and no one was certain how people got it. Of course, back then, making huge sums of money on a vaccine wasn’t a factor like today. Anyway, thank you so much. I look forward to reading your future posts.