One wonders what sexual favors were exchanged for the normally well-regarded publication Nature to run a defense of the indefensible, in this case, an obviously half-baked idea for Seattle residents to volunteer themselves for a home science study by jamming swabs through their noses to the back of their throats to collect their own Covid-19 samples and send them in by mail. Remarkably, the FDA woke up, cleared its throat, and said it needed to evaluate this “do it yourself” specimen collection.
Now mind you, that’s actually the more charitable interpretation as far as the Gates Foundation and the FDA is concerned. It is possible that the Seattle testing initiative was using mere nose swabs (as in just of the nasal cavity), which is not the current standard for Covid-19 tests. If that is so, the Gates Foundation and its allies in the Seattle medical-academic community are setting out to promote tests that are known to be less accurate via inferior sample collection. They will endanger patients and the public by virtue of generating false negatives due to choosing inferior sampling methods.
Yet Nature wails over the FDA stopping a testing program that either way was sure to generate a lot of bad samples and therefore bad results in Scientists baffled by decision to stop a pioneering coronavirus testing project.
Honestly, any scientists who are “baffled” are revealing their lack of powers of observation and common sense.
So what accounts for Nature shilling for such a flawed program? The too-obvious explanation is Bill Gates’ influence. He is the big money behind this so-called SCAN.
Before we take apart the Nature story (actually two but the second one is more important), it goes without saying that the US has a problem with Covid-19 testing, that in general, not enough is being done, and those who are worried that they might have contracted it don’t want to show up in an emergency room or outpatient clinic and risk getting infected. And medical professionals probably aren’t so keen about handling Covid-19 tests interspersed with other duties, particularly if they don’t have the best PPE to begin with. The same conundrum exists with going to your regular MD’s office to get tested (assuming you have one and assuming he’ll even test you).
However, South Korea already solved this problem: drive up testing. They also have walk-in testing stations, well enough staffed that there is no wait. Even in the supposedly backwards Deep South, we had drive up testing here in Birmingham, 20 minutes from where I live, up and running less than ten days after schools closed. I’ve seen more sites opened since then.
And I have not seen how the US version of these set-ups work, but in South Korea, all of the medical techs are hazmat-suited up, so they look well protected. As they ought to be in the US.
And here is why you want medical professionals to gather those samples. This is how the swabbing is supposed to be done:
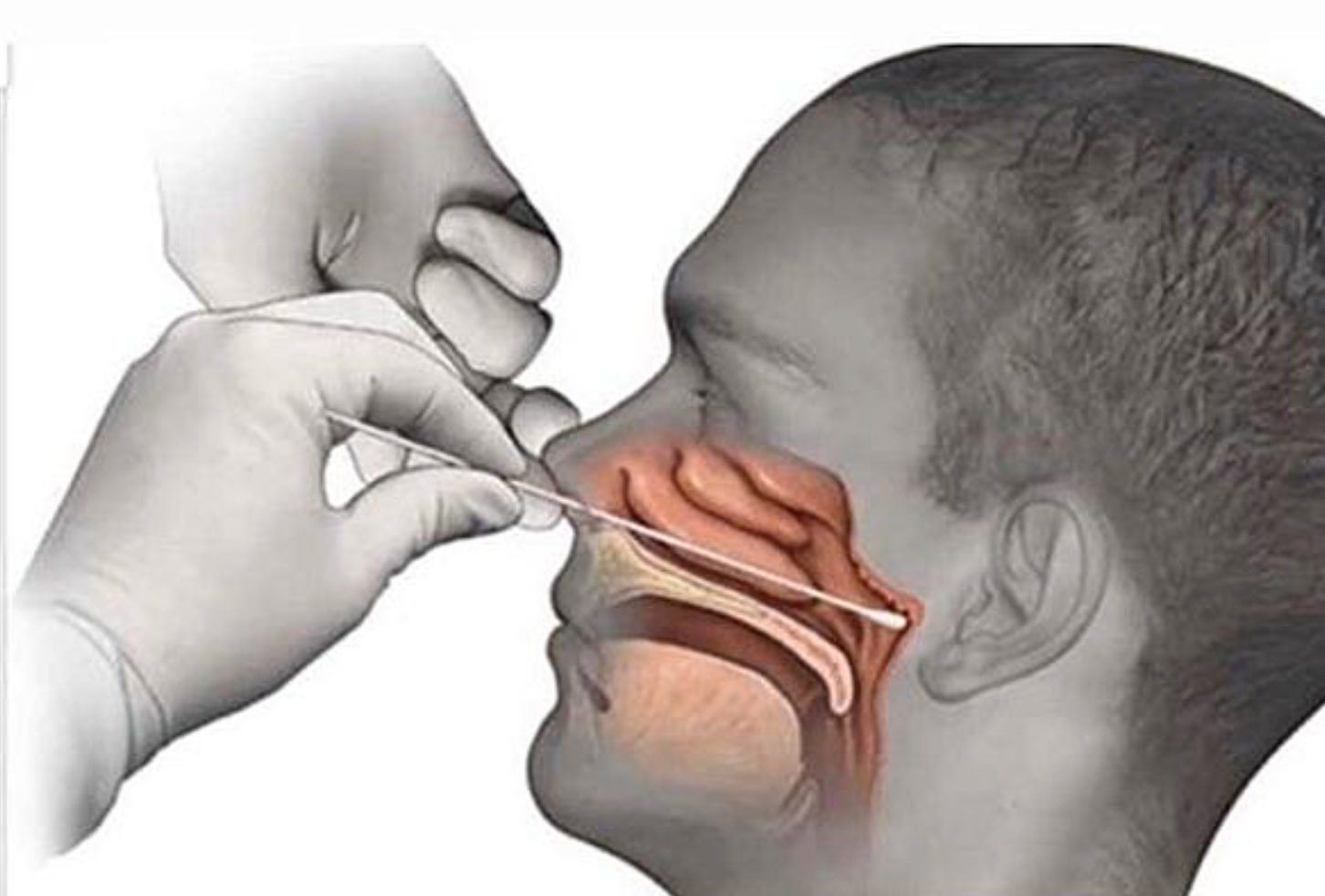
In other words, this “nose swab: which is more accurately called a “nasopharyngeal swab” is very invasive, as this Chicago Sun-Times story confirms (emphasis ours):
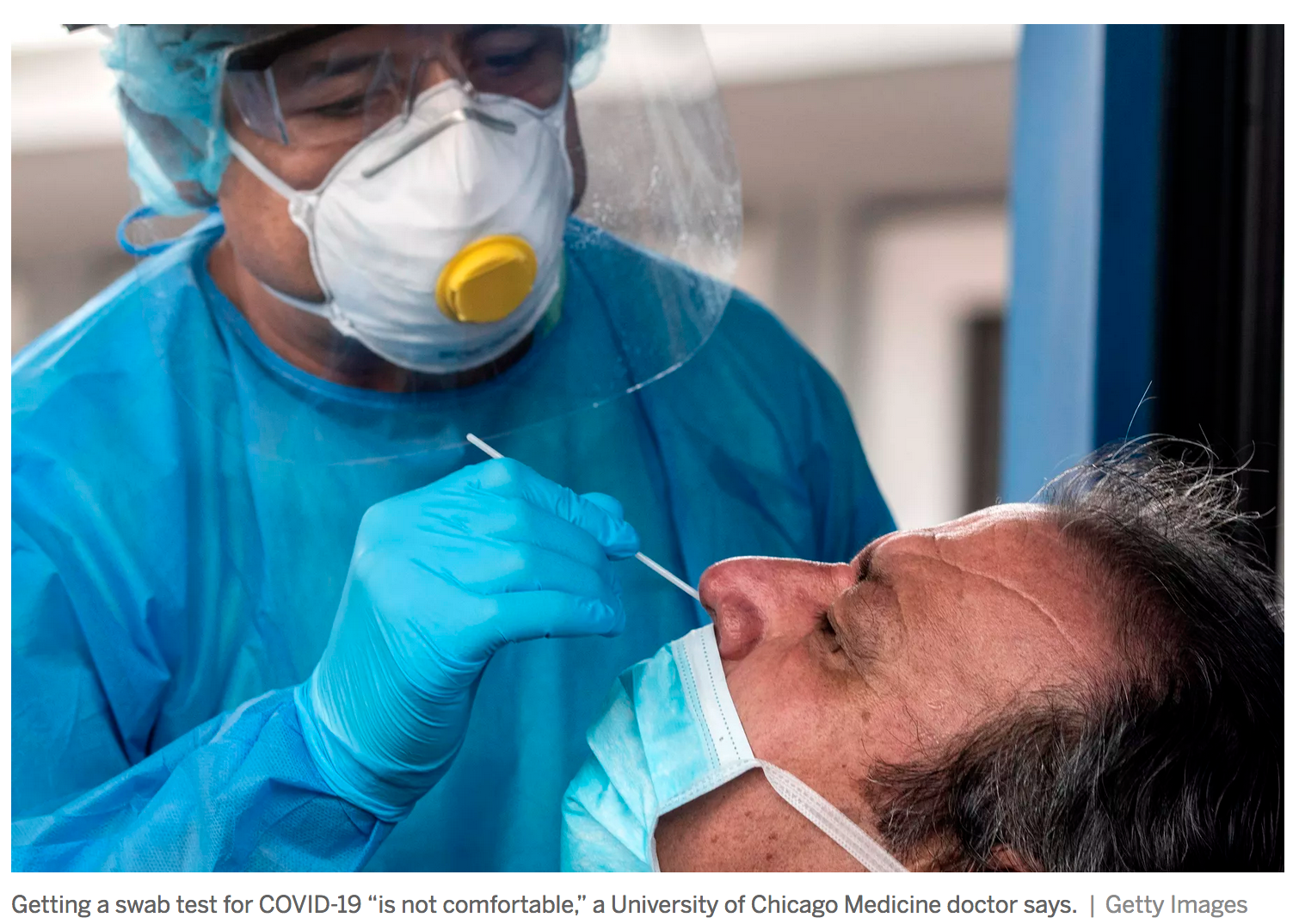
It isn’t pain like you feel from a shot. Instead, think about how it felt if you’ve gotten chlorinated water up your nose in a swimming pool.
The complex nerve endings in your nose told your brain: That’s not right, there’s something bad in there. And that burning sensation makes you want to blow the water right back out.
Well, the swab test for COVID-19 is more intense than that.
“It’s a deep burning, and it often elicits tears and sometimes coughing,” says Molly K. Erickson, a nurse practitioner at Rush University Medical Center who helped set up the Chicago hospital’s testing sites. “That discomfort really comes with getting an adequate sample.”
Got that? The test is nasty because it has to be to do the job properly. The nasty bit is hopefully short lived but that isn’t a given. For instance, in Business Insider:
While momentarily uncomfortable for most, it was “excruciating” for Pauw, who already had swollen nasal passages and had to drive 45 minutes to Tacoma, Washington, to get one.
“It feels like being stabbed, to me, in a really sensitive place and then having it be twisted,” she said. “And then, they had to do it twice.”
Even when it was over, Pauw didn’t feel better. In fact, she and her husband had just gotten on the freeway home when Pauw passed out. Fortunately, she wasn’t driving. “That was a horrible experience,” she said. “That was the very worst day of it all.”
One reason this test isn’t easy to administer is 70% to 80% of the population has a deviated septum, which means for them, there isn’t a perfectly straight path from their nostrils to the back of their throats
.
And while the plural of anecdote is not data, I happened to grumble to a colleague about the idiocy of expecting people to jam nose swabs to the back of their throats to do their own Covid-19 sample collection. He gasped out loud. Within the last hour, he’d gotten a call from a friend who’d gotten the Covid-19 sinus jab five hours earlier. He was on the verge of tears, he was in so much pain. “How can I ice myself in there?”
Mind you, the SCAN project has taken some important steps. It did help address a bottleneck, which was a shortage of test equipment. As a March 6 Nature article described, apparently before the home testing part had been launched, how lab testing initiatives exploded in Covid-19 hotspot Washington state after the FDA on February 29 authorized some academic labs to run tests. The Seattle Flu Study, dating from 2018, where people who think they have a bug send in nose swabs for analysis.
If you look at this video and instructions on how to do a flu swab, the “preferred method” is to use a swab that goes just as far back in the sinus cavities.
This raises the question as to the integrity of the Seattle Flu study process. Did they use the invasive nasopharyngeal swabs, or the more patient-friendly nose swabs? Plus for flus, a study published by the NIH didn’t find a major difference in results between nose swabs and the more daunting nasopharyngeal swab. But the nasal swab had a mere 89% accuracy versus 94% for the nasopharyngeal swab. That seems significant, particularly when a deadly disease is in play.
Back to the Nature stories. Here’s the snippets on the Gates efforts from the early March account:
Some of this burden could soon be relieved as the Seattle Flu Study changes course. This week, the Bill & Melinda Gates Foundation in Seattle said that it will furnish the study with another US$5 million. People who feel ill but aren’t in dire need of hospital care can now order the swab kit from the study website….
Dan Wattendorf, who directs efforts to create biotechnology solutions at the Gates Foundation, hopes that people with bearable illnesses will stay at home and order the kit, instead of going to hospitals where they could transmit the virus — or catch it
Notice the reinforcement of the false dichotomy: testing at home or testing in hospitals. No consideration of drive-by tests, or the medical vans created by Arnold Schwarznegger and disbanded by Jerry Brown as part of a pandemic emergency response program.
A correction to the story notes that Bill Gates has long has had his fingers in this Seattle testing pie:
It also said that the Seattle Flu Study was funded by the Bill & Melinda Gates Foundation. In fact, it is funded by Gates Ventures, an investment firm founded by Bill Gates. The Gates Foundation has provided funds for the study’s extension to COVID-19.
So Gates has a commercial, not charitable, motive. Glad we have that clear.
You’ll see in the second story simply brushes off the key question: could these at home samples be adequate? The FDA has every reason to be worried. Unlike the original Seattle Flu study, whose main goal was to trace the spread of seasonal bugs through the Seattle area, here the stakes are much higher: do you have a potentially deadly ailment or not? Do you need to self-quarantine and monitor your symptoms?
And if you watched the video or read the instruction, proper sample collection isn’t merely getting that swab to the back of your throat and removing it. The med tech is supposed to rotate the swab (up to five times!) and hold it in place five to ten seconds.
The May Nature story explains how the SCAN group exploited a loophole:
The FDA’s request to pause testing on 12 May frustrates many researchers developing diagnostics that can be conducted partially or fully outside hospitals…..
The researchers assumed that they were in the clear because the FDA had granted Washington state the ability to authorize emergency use of COVID-19 tests. In February, FDA guidelines precluded at-home testing from this type of provision, but made no mention of at-home specimen collection, which is how SCAN operates. As of 11 May, however, updated FDA guidelines do specify that tests with at-home collection need to be assessed by the agency.
It’s not clear the FDA has focused on the swabbing issue. It is concerned about samples possibly degrading via getting hot in transit. Having said that, the FDA has approved three “home based tests.” However, looking at test video from Labcorp, which has one of the “good to go” tests, it really is a nose swab, not a nasopharyngeal swab. So the FDA has signed off on a significant sacrifice in accuracy for the sake of convenience.
Back to the Nature account, which features the worst sort of “assume a can opener” thinking:
Robin Patel, president of the American Society for Microbiology, who is based at the Mayo Clinic in Rochester, Minnesota , says that researchers have long called for diagnostic tests to be deployed outside of healthcare settings. “This push is being accelerated now by COVID-19, because the reasons to do this are so apparent in an outbreak.” A model that requires health workers to draw samples can’t easily be scaled up. It’s labour intensive, and is risky for the workers required to stick long swabs up people’s noses.”
Yes, and it would also be nice if patients could draw their own blood so as not to expose health care workers to them either. Help me. How about investing in Ebola-level PPE instead?
So the answer is defective sampling and people getting false negatives because they didn’t swab adequately or contaminated the sample?
The article unintentionally makes clear that the vetting of the SCAN process didn’t check the accuracy of home-brew samples versus med tech collected samples:
In late February, the SCAN team cleared a first set of regulatory hurdles with the US Centers for Disease Control and Prevention. Next, the group analysed about 30 specimens with its own test alongside other assays in clinical labs, to vet its accuracy. With the data from these experiments, Washington’s department of health authorized the researchers to screen people and return the results to them.
In other words, the samples were assumed to be hunky-dory.
And it looks as if Nature completely missed the real story in lining up to defend Gates: that the FDA is approving home tests with nose swabs, which may indeed be what the SCAN tests were using, which studies have found to be markedly less accurate that the obnoxious nasopharyngeal swab. The result will be false negatives, meaning people who are sick will think they aren’t, and will infect others and possibly get quite sick before they get a second test and find out they have Covid-19. And all for the sake of Gates and commercial lab profiteering rather than staffing up and suiting up techs to conduct drive up and other more local tests.


Nature is pimping Gates funded nonsense. Lancet is asking Americans not to vote for the orange one. What’s wrong with science publications?
Gates is the new Armand Hammer/ George Soros, now?
Match for that straw? The post said no such thing. But Gates most certainly likes to throw his weight around, as I have heard from people who were party to his negotiations with developing countries about Gates Foundation donations.
The issue here is that this Seattle testing initiative has problems with sample collection no matter what approach it is using. And the FDA has approved at home testing programs that use nose swabs that have have been found in studies to be meaningfully inferior to the method used in hospitals to test for Covid-19, the nasopharyngeal swab. Yet Nature took no interest whatsoever in this issue. It appears to be due to deference to Gates and the academic institutions involved in this effort.
Gates has done a lot of good charitable work on third world health issues. However, even Nature pointed out that a private Gates venture is helping to fund SCAN.
Yves, can you help with this article? It seems that the creator of the PCR (Polymerase Chain Reaction) method was completely horrified at its use as a clinical diagnostic test. He had some harsh words for Fauci, too. I always hate learning one more time/way that Gates buys what he wants. I have lived in Redmond, WA for 35 years. Shame on Nature. Shame on Bill..
https://uncoverdc.com/2020/04/07/was-the-covid-19-test-meant-to-detect-a-virus/
A note to readers of SKBurns’ link, this is a quote from the article to give you a sense of what unbridled paranoia and membership in Qanon will lead to.
…” The people who have taken all your freedoms away in recent weeks, they’re social engineers, politicians, globalist thought leaders, bankers, WHO fanatics, and the like. Their army is composed of “mainstream media,” which is now literally a round-the-clock perfect propaganda machine for the Gates-led Pandemic Reich.”…
It doesn’t matter who stumbled on the properties of DNA polymerase isolated from thermophilic bacteria or what they think about it’s potential usefulness in amplifying segments of DNA in a mechanical thermocycling environment. Its a biochemical system that replicates any stretch of DNA bounded by synthetic nucleic acid primers specific to the DNA sequence of interest. It doesn’t matter if the DNA is from an unknown virus or used to determine the phylogeny of a novel plant species.
What is sad/comical about the author of the article is that they begin by saying they were trying to “cast off layers of ignorance”. Never underestimate our capacity for self delusion.
>Its a biochemical system that replicates any stretch of DNA bounded by synthetic nucleic acid primers specific to the DNA sequence of interest. It doesn’t matter if the DNA is from an unknown virus or used to determine the phylogeny of a novel plant species.
That’s exactly the point dumb dumb. PCR is a research tool not meant for diagnosing.
‘One wonders what sexual favors were exchanged’…that got my attention. You win the HL Mencken prize for today. ‘Nature’ as one knows is not just any zine, it’s serious science™. I’ve kept 1/2 an eye on what Bill’s been up to. I think any kind of at home kit is a bad idea, because the data on infections is pretty lousy as it is. There are many ways this kind of testing can go wrong with fairly predictable responses to that data and all this seems to be happening. More chaos, more confusing, more bad policy built on bad data. After a test like this a doc or the like should discuss the results with you. And that could result in other conditions being found… This type of testing, not something I’d do.
We all get some more Gates up our Noses?
He’s been getting up my nose for years with needless changes to the windows GUI.
Not to mention the outright spying and surveillance built into Windows 10, much of which can’t be disabled. Compared to everything Microsoft knows about what happens on your PC, a nasopharyngeal swab is temporary and maybe slightly less invasive.
Oh yes indeed. This ineffective and dumb thing has already happened here in the Tacoma area, Pierce County, just south of Seattle, King County. On May 10th The News Tribune reported this: Testing fiasco shows Western State Hospital COVID-19 response needs close scrutiny
“According to the Associated Press, WSH staff were herded into a small building, given test kits by people without gloves and told to “swirl a swab inside their noses.
A WSH forensic psychologist told the AP the testing procedure “put us all at risk” and no doubt yielded inaccurate results because the kits were designed for patients already exhibiting symptoms, not for asymptomatic carriers.
Keep in mind the state’s largest psychiatric hospital, run by the state’s Department of Social and Health Services, can’t hand out applesauce without adhering to strict enforceable codes, so why were they allowed to stick ineffective Q-Tips up staff’s noses without proper protection?”
For some reason, my colleagues at work(in academic biomedical research) love Bill Gates and see him as some kind of do-gooding angel that is above criticism. I have tried to discuss this with them civilly but they just won’t have it. In their eyes, he can do no wrong, I cannot figure out why, and they won’t explain why and seem to think I’m dumb for not thinking the same.
Clearly none of them is a GNU/Linux user! Anyone involved with the free software movement learned to distrust and detest Gates many years ago.
Ha! Maybe that’s my problem-I’ve spent plenty of time over the years with Linux users & software engineers. Look beneath the surface…there’s always an angle.
Having been up against Bill Gates and Microsoft since around 1980 and hearing the stories as well as suffering directly from both the business and technological practices, it has always shocked me to hear about what a noble and wise fellow Gates is supposed to be. I suspect this myth is the result of a well-funded and well-directed PR job which was embarked upon because Gates and his entourage came to understand that he and his works had developed a set of political problems — governments were looking into monopoly prosecutions, and many, many technicians had developed a vigorous hatred of Gates and his works and did what they could to encourage other vendors and business solutions when they could. I think if one investigates the beliefs of the believers one will discover the works of amply paid public relations.
This. Remember back before Gates was reborn as a philanthropist, and other billionaires were grumbling how Scrooge McGates was giving them a bad name? Then — hey presto! — PR was secured, the evil old Microshafty Gates was cleansed and the Brave New Gates was born, ready to cure the world of malaria, etc. etc.
I do hope that by the time I have to be tested there’s a sufficiently sensitive alternative test that allows me to avoid the nasopharyngeal swab.
I don’t think I’d volunteer for testing right now without a very high index of suspicion.
There’s no treatment for COVID-19 until you’re sick enough to be admitted to the hospital. Since that’s true I don’t see much value in being tested. I don’t plan to seek healthcare until I’m seriously oxygen deprived and in danger of my life. No testing for me until I stagger into the ER. Fortunately I already WFH full-time so I don’t need a diagnosis to stay away from people. I’m also decades younger than most of the people who die. God willing I’ll just tough it out. My experience of the US healthcare system involves hefty co-pays and a pitiless time-sucking insurer who denies every claim. I wasn’t born this way, I was trained.
Given the utter failure of any locality in the US to contain the virus we should all assume we’ll eventually get infected. Plan accordingly.
One has to wonder if all the elective procedures that were canceled this spring will put a noticeable dent in the usual number of iatrogenic deaths this year.
My job is in healthcare and I think a test might be necessary to be eligible for my 2 weeks paid COVID time. But my employer and the Canadian government keep changing their criteria so who knows.
I don’t expect to catch it until next fall at the rate my city is going.
Dont worry guys! Biden’s coming soon! He’ll keep gates in check :D
Waiting for a Facebook share of article with headline something like “Trump’s FDA bans covid testing” in 3, 2, 1 …
A friend in seattle did the SCAN test and it was a nasopharyngeal swab. Wonder how many kits weren’t returned once people realized what the test involved.
I know someone who was tested back in mid-March. She didn’t describe the details of how it was done, other than saying the test was performed by a physician assistant wearing hazmat gear.
Nine days later, she was informed that her test results were negative. Whew.
But not whew.
Since mid-March, I have noticed quite a change in her behavior. She sounds like she’s afraid to step off her property. Even to the point of taking walks around the neighborhood, which I think would help her quite a bit.
I think the test — and the lengthy wait for results — have traumatized her.
I had it done a couple of years ago by a nurse as part of a flu study. It was quite sharply unpleasant. The ‘water up the nose’ swimming pool analogy is accurate – my eyes instantly started watering and it was a struggle not to cough, contort my face, or otherwise try to dislodge the invasive object my brain told me was in there. Fortunately my nurse was very good, described exactly what to expect and how it would feel, and was in and out and done very quickly. It was still some time before things felt right again. It sounds like my reaction was on the mild end of the scale, but it’s still not an experience I am eager to repeat.
It is certainly not something I would want to attempt myself and I doubt that medical professionals would either. I’d rate it about on a par with taking a cornea swab, except that the defensive reaction occurs after the fact rather than before. In either case it would severely interfere with completing the procedure quickly and efficiently.
The video in this article is pure b.s. The swab is only about half-way as deep as necessary. See this one to appreciate how deep it has to go: https://youtu.be/DVJNWefmHjE
Wow, thanks!
Yes, it looked odd to me, but it didn’t occur to me to challenge a supposed instructional video by a medical products company. Guess it’s hard to find a model who won’t wince if the swabbing is done properly. The woman in the NEJM vid was a tough cookie but she looked apprehensive.
You would have to hold me down and probably sedate me to do that sort of invasive test! In fact the one time I did have to have a doctor put something up my nose I was sedated. One of the worst experiences of my life to that point.
And now you have Walgreens jumping into the testing arena by having you go thru a questionnaire with their lab partner and if they qualify you for the test, they then tell you which Walgreens to go to and a pharmacist guides you thru doing the nasal swab in your car while they stay safe in the building. Seriously, after reading this article you have to wonder how reliable the swab will be and whether or not you’ll get an accurate result.
“The only current COVID-19 specific data comparing OP with NP comes from two low quality, non-peer-reviewed studies and should be viewed with caution. It is not possible to accurately assess sensitivity from the existing data and there are no data to assess the diagnostic impact of combining both tests.
We found two COVID-19 studies, one with 213 patients and 205 OP and 490 NP samples (1) and another with nine patients and an unknown number of samples (2). The smaller study by Wolfel found no difference in detection rates between OP and NP but Yang, the larger of the two studies, reported OP swabs detected the COVID-19 virus less frequently than NP swabs and should not be used in place of NP swabs. This difference was most notable at days 8+ after illness onset, with about a 20-percentage point minimum difference in positive rates between OP and NP swabs. The difference was less at 0-7 days, with about 60% COVID-19 infected patients positive on OP vs about 70% on NP.
Both studies recruited hospitalised patients and it is unclear whether the primer used in the RT-PCR was the same as used in other countries. Applicability to other healthcare settings is therefore uncertain. Overall, the data are not robust but we would caution against relying on OP alone over NP.”
https://www.cebm.net/covid-19/comparative-accuracy-of-oropharyngeal-and-nasopharyngeal-swabs-for-diagnosis-of-covid-19/
So the comparison is between 70% specificity for NP that is impossible to self-administer versus 60% specificity for OP that can be self-administered. That’s not an inherently bad trade-off.
Is there any video that shows someone doing the nose swab on his-/herself? I mean, properly.
I doubt that it’s possible.
I had my first ear pierced by someone else as a boy of 15 and the second one professionally.
I performed the third ear piercing by myself, pushing the stud through my earlobe in front of a mirror.
It all went well, but my hands were shaking and I felt queezy afterwards.
I’m pretty shure, your body will keep you from doing the nose swab properly.
And also, I suggest we perform the nose swap on all those ‘baffled’ scientists and see
how baffled they are afterwards that anyone would want to do this to his-/herself.