By Lambert Strether of Corrente.
As readers know, I’m a big fan of anti-covid nasal sprays. The first time I did a round-up, in July of 2021, there were a few vaccines under development, but the focus was mainly on prophylaxis and treatment. As of this writing, there are many more solid nasal vaccine projects, worldwide, and one treatment on the market. So I’m encouraged!
I won’t go very deeply into the science behind the new generation of nasal vaccines (the previous post includes a diagram of the “human oral squamous epithelium” and how SARS-CoV-2 affects it). Rather, I will look at the advantages of nasal vaccines, ask how many nasal vaccines are under development (not so easy to answer), and look at a few of the projects in more detail. I’ll conclude with some brief caustic remarks on the Biden Administration’s miserably inadequate handling of this technology.
Advantages of Nasal Vaccines
Nasal vaccines have two significant advantages over current intramuscular (syringe-injected) vaccines.
First, the mjeans of administration means broader take-up.
Nasal vaccines don’t trigger fears of needles:
Trypanophobia is the extreme fear of injections or hypodermic needles. While it tends to be more common in children, this fear can remain well into adulthood for some people. Around 20% of adults in the world share this fear, with 1 in 6 adults avoiding the flu vaccine due to this phobia. If needles are keeping you from getting vaccinated against COVID-19, good news is on the way!
It’s always nice to have a word of the day!
Nasal vaccines are easier to administer:
Administration does not require a trained professional, sterile environment, and syringe and needle; making self-administration possible and thus offering much broader possibilities of distribution and a greater uptake in vaccination.
Self-administration is really attractive to me, since I avoid interacting with the health care establishment as much as I possibly can.
Some nasal vaccines can be easier to produce and store:
[Peter Palese, chairman of the Department of Microbiology at the Ichan School of Medicine at Mount Sinai Hospital] said one of the major advantages of [his] nasal vaccine is its ability to be stored in a normal fridge at 2-4 degrees Celsius, rather than the ultra-low temperatures required for the Pfizer and Moderna vaccines.
Because the vaccine is grown in chicken eggs — the same technology used for many flu vaccines all over the world — the cost of development is also cheaper. “It is much, much cheaper to produce this vaccine as compared to the mRNA vaccines by Pfizer and Moderna,” Palese told DW
Second, the mechanism of action promises sterilizing immunity. From the American Chemical Society:
SARS-CoV-2 often enters through our noses [because Covid is airborne], where it encounters a protein called ACE2, which is found in abundance in our nasal passages. ACE2 is the virus’s doorway into our cells. In fact, the mucosal membranes that line our airways, digestive systems, and reproductive tracts are often the first parts of our bodies to face an invading pathogen. A network of immune cells resides underneath our mucous membranes, or mucosae, and forms a front line of defense against invaders, and they prevent most infections from taking root. This is the mucosal immune system, and some immunologists think we have been seriously underestimating it.
“When you think about it, that’s where we acquire most of our infections: we inhale them, we consume them, or we get them through sex,” says Michael W. Russell, a mucosal immunologist and professor emeritus at the University at Buffalo.
Our mucosal immune cells make a special class of antibodies that are constantly secreted from the mucous membranes to protect the nose, gut, and other vulnerable sites from pathogens we’ve seen in the past. “But if you don’t stimulate the immune system in the mucosae, you don’t obtain mucosal immune responses,” says Pierre Charneau, head of the Molecular Virology and Vaccinology Unit at the Pasteur Institute.
Yet most research on SARS-CoV-2 and our immune systems has overlooked mucosal immunity in favor of the easier-to-study systemic immunity. “When the pandemic hit last year and I started to see papers coming out about immunity, it really quite staggered me to see an absence of attention to the mucosal immune response,” Russell says.
Charneau and a group of scientists in Paris have shown that natural SARS-CoV-2 infections trigger both systemic and mucosal immunity. But our current crop of COVID-19 vaccines offer only systemic protection. Developing vaccines that are sprayed up the nose, rather than injected into the arm, could change that, Charneau says. Mucosal immunity in our noses could be like a guard at the door, potentially helping stop even small infections of SARS-CoV-2 right where they start.[1]
Stopping infections where they start could mean sterilizing immunity. From the Lancet:
[Intranasal] vaccines have the potential to induce sterilizing immunity against mucosal pathogens. Antigens are exposed at the initial site of viral attack to induce potent immune responses at local or distant mucosa.
In a word, the [intranasal] immunization route can induce sterilizing mucosal and systemic immunity, further preventing virus infection and transmission. Application of [intranasal] vaccines will hopefully help deal with the persistent COVID-19 pandemic and potential viral contagious diseases in the future.
Time concludes:
[R]esearchers hope that nasal vaccines may one day do what even the highly effective mRNA vaccines made by Pfizer-BioNTech and Moderna have not: slow transmission enough to bring the pandemic to an end.
How Many Nasal Vaccines are Being Developed?
It’s not easy to determine how many nasal vaccines are in the works. I can’t browbeat Clinicaltrials.gov into giving me a figure less than 111, which is wrong. WHO says 9. The Lancet has a table that lists 12 (the figure used by the Times).
My guess is that the field is very dynamic, and the records-keeping hasn’t kept up. So let me just name a series of projects, so you can can see what I mean. One study, Altimmune, is known to have failed. Yale and Stanford have projects. Oragenics Inc. in Tampa, Florida and Inspirevax in Laval, Quebec have a hamster study. The Swiss National Fund has a phase 1 clinical trial, as do Sweden’s Karolinska institute, and Codagenix in the United States. France has a pre-clinical trial. Russia has a Sputnik spinoff. There are more projects in Mexico, at Washington University in St. Louis, and at Mount Sinai Hospital. I’m sure there are more; I could hardly turn around without stumbling over one. There are eleven mentioned here, plus three[2] I am about to list, which comes to fourteen, which is — allow me to take a moment and break out my calculator — two more than the twelve listed by the Lancet. Encouraging!
Examples of Nasal Vaccines
I want to delve more deeply into three projects in the United States, India, and Thailand.
First, the United States, at the National Institute of Allergy and Infectious Diseases (NIAID):
NIAID scientists have developed a candidate COVID-19 vaccine targeted for infants and young children that would require one dose delivered by a nasal spray.
In a new study published Dec. 7 in PNAS, NIAID’s Ursula Buchholz, Ph.D., Shirin Munir, Ph.D., Cyril Le Nouen, Ph.D., and colleagues in Bethesda, Maryland, and Hamilton, Montana, describe how they used a weakened version of a bovine/human parainfluenza virus (called B/HPIV3) to deliver SARS-CoV-2 spike protein to stimulate immunity against COVID-19. Think of how we use a grocery cart to carry food items to our vehicles. In this case, B/HPIV3 is the cart loaded with protective proteins that train our immune system to protect against SARS-CoV-2 and HPIV3. The vaccine is noteworthy because the nasal delivery creates immunity in the respiratory tract, which is thought to be particularly effective in restricting respiratory virus infection and transmission.
The group developed two versions of the vaccine for SARS-CoV-2 and compared them in hamsters to mimic COVID-19 in people. Candidate vaccine B/HPIV3/S-2P performed the best and was safe and effective at preventing SARS-CoV-2 from spreading in the nose and lungs.
OK, granted, a hamster study. But this is the first I’ve heard of it, and I do try to keep track. What a shame the director of NIAID has no clout! Because otherwise they surely would have brought this excellent result before the public;
Second, India, at pharma giant Bharat:
Bharat Biotech, the maker of the first indigenous Covid-19 vaccine Covaxin, completed phase 1 trials on 400 healthy individuals.
In November last year, it completed phase 2 trials on about 650 volunteers in 10 centres across the country.
In the last week of January, the Drug Controller General of India (DCGI) permitted the vaccine maker to undertake phase 3 trials. BBV154 is an intranasal replication-deficient chimpanzee adenovirus SARS-CoV-2 vectored vaccine.
The vaccine has been developed using a new technology licensed from Washington University. Bharat Biotech believes in a cocktail approach of administering two doses of two different vaccines using a combination of intramuscular and nasal[3] in a first of its kind approach.
“If successful, intranasal vaccines make the roll out of booster doses easier and faster as administering intranasal vaccines is much easier when compared to that of intramuscular vaccines,” said one of the directors of a city-based corporate hospital adding that any danger of the fourth wave of the pandemic be dealt with a comprehensive programme for a booster dose to all.
The trials are expected to be complete in the second week of April.
Finally, Thailand:
A nasal spray said to be effective in preventing coronavirus infection is being developed by Thai scientists, and the product will be ready for launch later this year, according to the developers.
The spray is being developed under joint efforts by Chulalongkorn University, Silpakorn University, Health Systems Research Institute (HSRI), the Government Pharmaceutical Organization (GPO) and an undisclosed private bioscience company.
Dr Chanchai Sittipunt, dean of the Faculty of Medicine at Chulalongkorn University, said a cooperation agreement has been signed to produce the anti-Covid-19 nasal spray, and it could be a subject for global research.
Now the technology is being transferred to the private sector, which is conducting clinical research to meet the standards needed for registration with the country’s Food and Drug Administration, he said.
Dr Chanchai expects the spray to be made available for sale in the third quarter of this year, adding Silpakorn University’s Faculty of Pharmacy has studied the spray with Chulalongkorn University’s Faculty of Medicine using lab animals.
This project is a little bit of a dark horse. Nevertheless, Chulalongkorn is a respected institution, and Thailand, which is facing the collapse of its tourism industry, has every incentive to get this right, not just for medical tourism, but all tourism.
Examples of Nasal Treatments
From the Economic Times of India:
Glenmark and its partner Canada-based SaNOtize Research & Development Corp. on Wednesday announced the launch of its Nitric Oxide Nasal Spray (NONS) under the brand name FabiSpray in India for the treatment of adult patients with COVID-19 who have high risk of progression of the disease.
NONS when sprayed over nasal mucosa acts as a physical and chemical barrier against the virus, preventing it from incubating and spreading to the lungs. Glenmark received manufacturing and marketing approval from India’s drug regulator for NONS as part of an accelerated approval process.
Glenmark said the Phase 3 trial in India met the key endpoints and demonstrated reduction of viral load of 94% in 24 hours and 99% in 48 hours, and was found to be safe and well tolerated in COVID-19 patients.
NONS has already received a CE mark in Europe, which is an equivalent of marketing authorization in case of a Medical Device.
By virtue of the CE mark, SaNOtize has permission to launch NONS in the EU.
NONS is also approved and being sold in Israel, Thailand, Indonesia and Bahrain, under the name enovid™ or VirX™.
So, world travelers, you can pick up a few boxes or a case of the stuff, eh?
Conclusion
One of the encouraging things about these nasal spray developments is they are either on the market already (NONS), or approaching the market (Bharat in India, and Thailand). Among the many, mahy heinous crimes of the Biden administration was not to accelerate the development of nasal spray technology. They had the example of the Other Guy’s Operation Warp Speed before them, and yet they sat on their hands, or diddled. It makes little difference, at this point, whether their indifference was from malice, or not. In either case, the endpoint was denial of life-saving medication to the citizenry they betrayed. It’s almost like they don’t want the pandemic to end.
NOTES
[1] Yes, but is that a sustainable business model?
[2] Bharat licenses Washington University’s technology, so perhaps my definition of “project” doesn’t line up with the Lancets. Ah well, nevertheless.
[3] Wrong. This is Yale’s approach.
APPENDIX 1
Here is Google:
No hits, but the ad is a very nice touch.
Here are the results I was looking for, because I knew they existed:
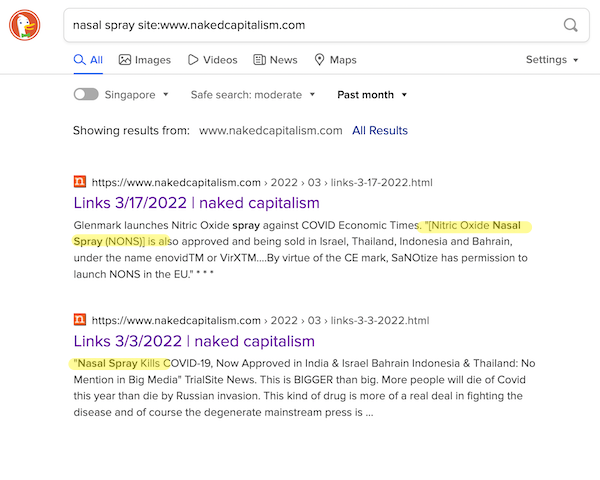
It’s pretty hard to argue this isn’t outright censorship from Google. Badge of honor, so far as I’m concerned.
APPENDIX 2
Speaking of nasal sprays as prophylactics, the FTC isn’t happy with Povidone. But the Povidone market is expected to do very well.


I get a couple hundred results if I go to one of the supposedly unattributed country searches from
https://ambition.dk/impersonal/
and use
[site:nakedcapitalism.com nasal spray]
I also get results from a cse
Thank you!
That entrance to Google’s search engine finds some old writing of mine I thought lost, that didn’t show up at all at the front entrance.
But that means that Google’s algorithms hasn’t just gone bad, and doesn’t just sort the hits you should want at the top, it actively hides the “wrong” results based on who they think you are. That is mindboggling.
Maybe Google just doesnt care enough based on who you are in some circumstances to put the resources into hiding things?
Thanks much for that pointer, Kevin. I’ve also given up on Google and mostly use Brave or occasionally DDG. The lack of an honest broker for search is annoying, to say the least.
NC uses a SQL database, I presume. Why is it not possible to directly perform simple searches like this? It may take a DBA to set up indexes for the relevant tables, but this is basic, known tech.
> NC uses a SQL database, I presume. Why is it not possible to directly perform simple searches like this?
Because our comments database is enormous and it would take real work beyond indexing to unhose it (possibly even rewriting the queries against it, which seem suboptimal).
Yes, doing a targeted search of comments will probably require writing queries and adding a page to the site to facilitate the search.
But again, this is not rocket science (e.g.,: SELECT * FROM comments_table WHERE comment_text LIKE ‘%string%’).
Surely there are professionals in your network or even the NC commentariat who could help with this?
I’m available!
It gets much more interesting with a very large database that isn’t indexed. As in, your hosting will charge you an interesting amount for CPU cycles.
That’s one of the reasons why adding an index would be a good idea, though the choice of the word ‘unhose’ suggests there might(?) be some other issues.
Basically, it irks me to see Lambert having to deal with the censorship and cr*p search results from Google and DDG when it should be possible for him to directly search the NC database. That is, even if NC doesn’t provide a public interface for us visitors to search comments (e.g., for reasons of cost), I would think a private search for Lambert, Yves, et al, should be possible.
I thought NC runs WordPress, in which case everything comes from a typical SQL database that includes a table called wp_comments. I assume that each time we load a web page, WordPress performs a SELECT on wp_comments, gathering up and sorting all comments associated with a given page. A wider search of all comments would be just a different SELECT query. In other words, yes, it would be necessary to write a query and do a bit of coding to make that available, but again this isn’t rocket science.
The big search engines have sold out, gone woke, gone evil, whatever. We can’t count on them any more for unbiased, unfiltered results. For this one site, though, NC evidently has its own database of comments and articles, and I see no technical reason why it couldn’t be searched directly.
As much as I think they don’t like people telling them to do stuff, I’d be in favour of a new $50,000 fundraising goal to make this happen.
Pulled that number out of my ass. Basically whatever it would cost to do it and pay someone somewhat above market rate. Maybe $100k?
Thank you for putting this together! It’s a tiny little blip of encouragement in a very unencouraging time.
More than a blip. It’s about to become a steady drip!
This is very good news. If they provide sterilizing immunity, it’s huge.
I haven’t even taken the flu shot this year. The previous two years crippled my trust in the healthcare industry.
If this pans out and another deadly Covid variant shows up, I might bite.
As long as they don’t turn it into a status symbol and threaten jobs over it…
I’ve done a little reading about flu vaccines and it seems that if you get a shot every year you are less protected than if you get one every 2-3 years. Perhaps getting flu vaccine annually inhibits your natural immunity. At any rate, at best the flu vax has an efficacy of 40%.
https://www.verywellhealth.com/flu-vaccine-effectiveness-5211793
If the NONS “acts as a physical and chemical barrier against the virus, preventing it from incubating and spreading to the lungs” could it also work as prophylaxis? If it’s safe to use daily, sign me up!
University of Melbourne
Heparin based vaccine
https://about.unimelb.edu.au/newsroom/news/2021/december/nasal-spray-to-fight-covid-19-heads-to-clinical-trial
I personally would be wary of a spray containing Heparin.
Very well done, Lambert! Thank you. This is certainly the way to go regarding vaccines…and has been since the beginning. But it’s so, well, old fashioned.
What about povidone iodine as a nasal spray? You mentioned it in passing at the bottom. Is it something you have looked e looke into and rejected? https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8026810/
FredW – Povidone has been recommended on NC several times over the past two years.
IIRC, it was Yves who first mentioned it in a post. Several commentators, including me, have also recommended it.
No one will do a clinical trial on it because it’s not patentable. You’ll at most get some underpowered studies.
A general summary on Povidone as a viricidal for Dentists (but recommended more widely for medical workers)
The use of Povidone Iodine nasal spray and mouthwash during the current COVID-19 pandemic may protect healthcare workers and reduce cross infection.
https://www.nature.com/articles/s41415-020-2402-0
Linked to at NC on 8/7/2020 (the SSRN version).
See also Povidone‐iodine gargle as a prophylactic intervention to interrupt the transmission of SARS‐CoV‐2, linked to on 8/5/2020.
And see also Povidone-Iodine Demonstrates Rapid In Vitro Virucidal Activity Against SARS-CoV-2, The Virus Causing COVID-19 Disease, linked to on 8/9/2020.
An Australian study is being conducted starting next month (I would have volunteered but I live alone and the study appears to be monitoring household transmission)
original paper mentioned in the article: https://www.theajo.com/article/view/4466/html
Heparin spray as mentioned by Barry above and which I shared a link to a few months ago also notable, and promising I hope!
Woop! Woop! Woop!
I need to sound the alarm on this topic. Here’s why:
For around three months, I was using povidone iodine prophylatically. In order words, I was doing at least two squirts per nostril per day.
In mid-January, my nose started running nonstop. It was as if I had a faucet on my face.
At first, I thought that I had low-grade symptoms of you-know-what, but I wasn’t feeling feverish or fatigued. But I did kick the nose hosing into high gear. Two or three squirting sessions per day.
By early February, my runny nose problem was abating. I figured things out and concluded that I was probably experiencing hay fever. Plants bloom early in Arizona, and this year has been off the charts as far as pollen is concerned.
In late February, I started taking an herbal remedy, freeze-dried stinging nettles. It’s good for alleviating the symptoms of hay fever, and, over time, it will help your body resist the underlying pollen allergy.
As mentioned here previously, I’m a huge fan of the FLCCC, and I tune in to their Weekly Updates on Wednesday afternoons. Sometime during late February or early March, Dr. Paul Marik warned against the use of povidone iodine unless one has been exposed to COVID.
A few weeks later, one of the nurse practitioners who was answering audience questions during the Weekly Update noted the presence of elevated levels in the blood of people who’ve been using a lot of povidone iodine. I presume that those elevated levels are iodine, but the NP didn’t say. But the NP did note that such elevated levels can lead to thyroid problems.
So, being forewarned, I have stopped using povidone iodine on a daily basis. I put it in the medicine cabinet — on the “just in case” shelf.
Thanks for this heads-up warning about using prov. iodine on a daily basis.
You’re welcome! Happy to be of service.
I wonder if this isn’t why the label on Betadine says not to use for more than 7 days.
From the viraldine.com website:
Over The Counter povidone and betadine contains scrubbing additives and fragrances that shows through clinical trials leads to mucosal damage & causes bleeding with short or long term use. Our PVP-I comes in powder, pure form and is compounded using only natural ingredients.
An aside: The slightly garbled English there does not inspire confidence in their product.
Lambert: It’s pretty hard to argue this isn’t outright censorship from Google. Badge of honor, so far as I’m concerned.
It would seem that you are in good company (although this article pertains to social media, not Google per se):
Algorithm-ranged newsfeeds were introduced as a way of helping users avoid missing out on information they consider important amid the endless stream of news. At the same time, as tech giants have taken control of what users see first, they have faced accusations of biased moderation.
“If you’re saying something that is just wrong, we don’t take that down, but we stop it from spreading, generally. That’s a much more sensitive topic,” said Zuckerberg.
https://sputniknews.com/20220328/questions-still-unanswered-about-possible-biased-moderation-on-major-social-media-platforms-1094248386.html
The important thing to the gatekeepers like Goog and FB and Twit is not accuracy or correctness in what they “fact” check; the important thing to them is keeping the power they’ve given themselves over speech. That’s it, that’s the entirety of their goal. Exercising power is their way of keeping power; exercising that power arbitrarily increases their power. imo.
And if you’re going to kick a bit of sand at Google, you can save this for when you feel like kicking the Gray Lady in the nuts:
New York Times’ Fear of Ordinary People Talking Back
The readers, and their reactions, are everywhere. They’re in the comment section, on Reddit and on Twitter. They know what you look like, and they know how to tag you on social media when denouncing your last article. Unlike the typewriter clackers of yore, today’s journalists instantly hit publish, and within minutes their articles are torn apart on social media, both a sign of our advancing technology and the consequences of living in a free society.
https://fair.org/home/new-york-times-fear-of-ordinary-people-talking-back/
Many kudos and thanks for this post, Lambert. Such a spray would literally be such a lifesaver, a complete game changer.
And shame on Google search — gaw. “Don’t be evil.” Try: “Don’t be useless.”
I hope I didn’t miss this info sitting right out in the open, but do we have a solid idea of how long sterilizing mucosal immunity might last?
In one of the articles, the comparison was made to natural SARS2 infection which we know doesn’t produce very long lasting protection from reinfection. Seemingly less and less with the recent variants.
So I would assume that to make a big crack at local or regional elimination of the virus, we’d be looking at needing to get a large ℅ of the population vaccinated nasally all at once and probably boost every 3 months for a while. This seems depressingly unlikely to happen, even if the authorities went all-in on this strategy. I imagine many more people will oppose mandatory vaccination Part 2.
Hopefully there’s an element here that I’m failing to consider. Thanks so much for this roundup!
This site claims to sell NONS (Enovid) and ship to the US
https://buyenov.com/a/faq?gclid=CjwKCAiAjoeRBhAJEiwAYY3nDBQtbRB9UEbSJogojSRak7Hlco-Pz3YuTymlw3VPiJb31p9OtDTSHRoCu_0QAvD_BwE#AbouttheProduct-5
I would want to make 100% certain that the vendor was legit, e.g. a licensed pharmacist I physically went to.
I’ve been using Enovid for about a year, I order it for the whole family. Very good service from IsraelPharm. Not endorsing, just FYI – in the beginning I got it from Ebay but this is must cheaper and faster
For influenza there is a nasal spray vaccine called ‘Flumist’ developed by Medimmune which is now part of Astra Zeneca. It is using live attenuated virus and is as effective as the traditional vaccines. It is not nearly as popular as the traditional flu vaccines though.
I would use the word that sometimes it is effective as the traditional vaccine. It is also a lot more expensive and is not approved for use in older adults.
Having worked in pharma I have learned don’t get too excited about early clinical trials or preclinical especially when it is done on rodents. There are many cures in rodents for a wide variety of diseases that do not work in humans. Real studies take take and lots of money.
According to discussion on Swedish television -. I think regarding the Karolinska study – immunity in the nasal area last in general less time than when the whole body is treated.
So yes (from that snippet, one can always hope in this case it lasts longer). Then again with an immunising vaccine, there may be new, more localised strategies that might make sense.
So MedTwitter (as well as Zoe in the UK) are reporting high frequency GI symptoms with BA.2 – mimicking Norovirus and in some cases probably is. That would suggest perhaps we are back to caution around fomite transmission, and the question could be raised of whether there is value-added to the nasal mucosa approach. Not saying its not still an airborne virus, but what if respiratory entry is sometimes secondary?
Thanks, Lambert, for this information about nasal sprays. I phoned my local pharmacist and he has not heard anything about a nasal spay that fights covid.
If the spray works well in preventing disease then the price according to the website is also good. Each package has 200 spays. It can be used before you go to an indoor setting or after you have left such a place in order to keep the virus from infecting your lungs.
Love the symptomatic cognitive dissonance packed into that Time quote:
Homage to mRNA vax is de rigeur.
This:
https://www.immunoprecise.com/immunoprecise-partners-on-launch-of-sars-cov-2-nanomedicine-therapy-program/
is to me the most interesting approach that I have seen, and I’ve seen enough to look askance. Nanobody VHH’s are the latest once and future king betrayed by this mRNA mass experiment usurper. A pity not enough Project Lightspeed coin has been thrown towards this project which has sound science backing it.
This looks to be yet another. Results announced yesterday claim it’s effective against all COVID-19 variants in mice.